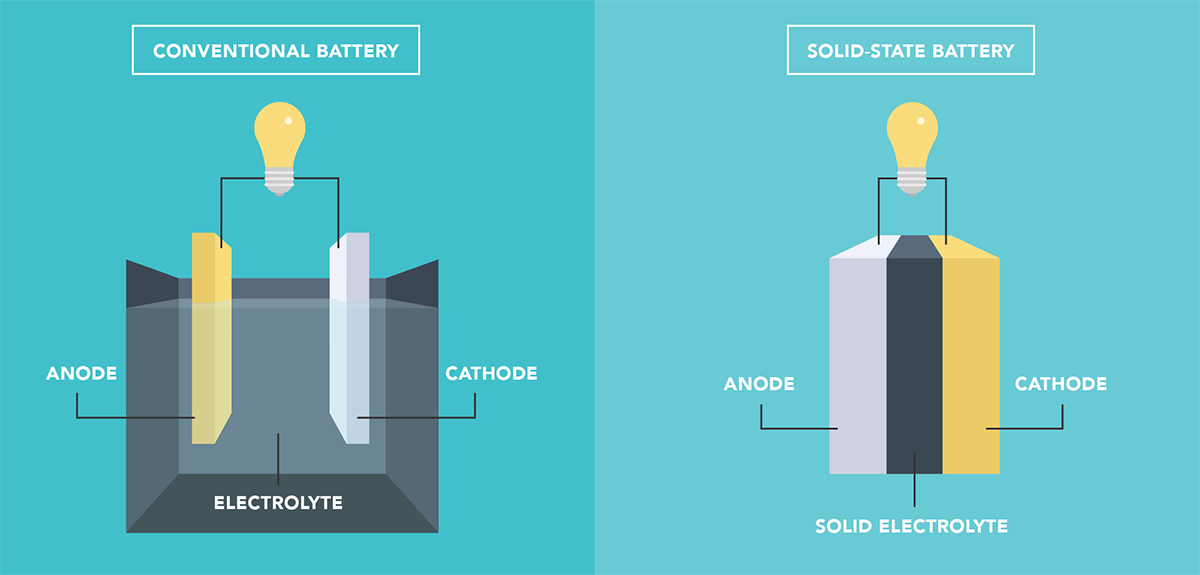
Researchers from the Illinois Institute of Technology, Argonne National Laboratory, and University of Illinois at Chicago say they have created a room-temperature solid-state lithium-air battery that is rechargeable for 1,000 cycles, has a minimal polarization gap, and operates at high rates.
In “A room temperature rechargeable Li2O-based lithium-air battery enabled by a solid electrolyte,” published in the journal Science, the researchers explain that their Li-air batteries use a four-electron redox mechanism with a composite polymer-ceramic solid-state electrolyte. Senior author Mohammad Asadi said the battery design could store 1 kWh/kg or more, four times more than current Li-ion batteries.
“A lithium-air battery based on lithium oxide (Li2O) formation can theoretically deliver an energy density that is comparable to that of gasoline,” claim the authors. “The composite electrolyte embedded with Li10GeP2S12 nanoparticles shows high ionic conductivity and stability and high cycle stability through a four-electron transfer process.”
Source: Illinois Institute of Technology, Science Journal via Green Car Congress