An international team of scientists, including researchers from the DOE’s Argonne National Laboratory, has discovered an anode material that enables super-fast charging and offers stable operation over many thousands of cycles.
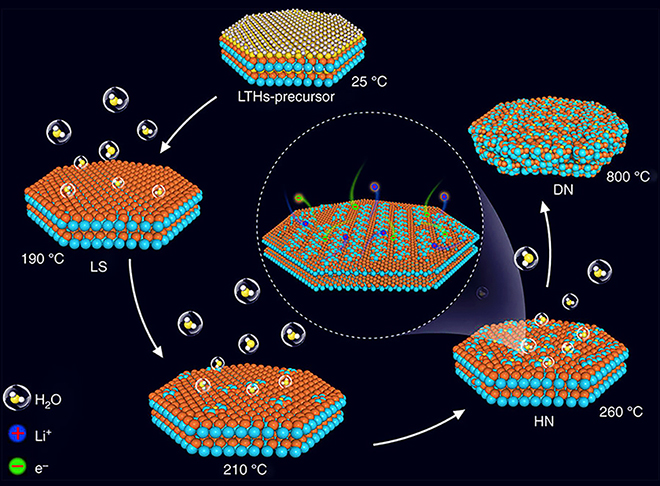
In a recent article in Nature Communications, Argonne Battery Scientist Jun Lu and colleagues describe a water-bearing compound, lithium titanate hydrate, that could replace the graphite anode commonly used in lithium-ion batteries. Past research had identified lithium titanate as a promising anode material, because of its potential for fast charging and long cycle life, as well as safer operation compared with graphite. In synthesizing this material, researchers used a water-based process that involved a final step of heating the anode material to above 500° C to drive out the water completely. This step was needed because, during battery operation, the water would react with the electrolyte and degrade performance.
Argonne Distinguished Fellow Khalil Amine, a co-author of the study, noted that heating to such a high temperature caused unwanted coarsening and clumping of the structure. The international team found that, by heating the anode material to a much lower temperature (less than 260° C), they could remove the water near the surface, but retain the water in the bulk of the material without coarsening and clumping of the structure. When the scientists tested the material in the laboratory, cycling stability improved and capacity degraded only slightly over 10,000 cycles. The material also charged very quickly – within less than two minutes. “Most of the time, water is bad for non-aqueous lithium-ion batteries. But in this case, it can be downright good,” said Jun Lu.
Looking to the future, Jun Lu observed that, because water is everywhere in nature and common in chemical synthesis, the fabrication approach reported in this research could open the door to discovery of other high-performance electrode materials.
Source: Argonne National Laboratory