In the quest for higher energy density, the use of lithium metal anodes would seem to be a promising approach, if it weren’t for those doggone dendrites. Dendrites are microscopic fibers of lithium, which sprout, tree-like, from the surface of the lithium electrode and spread through the electrolyte until they reach the other electrode, possibly causing a short circuit.
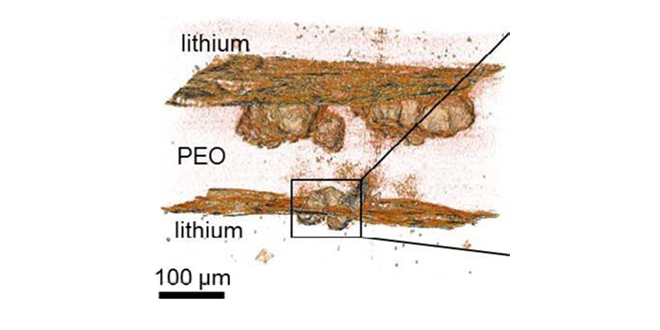
Researchers at the DOE’s Lawrence Berkeley National Laboratory are trying to get to the “root” of the problem, and have discovered that during the early stages of development, the bulk of dendrite material lies below the surface of the lithium electrode. Using X-ray microtomography, a team led by Nitash Balsara observed the seeds of dendrites forming in lithium anodes and growing into a polymer electrolyte during cycling. It was not until the advanced stages of development that the bulk of dendrite material was in the electrolyte. Balsara and his colleagues suspect that non-conductive contaminants in the lithium anode trigger dendrite nucleation.

“Contrary to conventional wisdom, it seems that preventing dendrite formation in polymer electrolytes depends on inhibiting the formation of subsurface dendritic structures in the lithium electrode,” Balsara says. “In showing that dendrites are not simple protrusions emanating from the lithium electrode surface and that subsurface non-conductive contaminants might be the source of dendritic structures, our results provide a clear prescription for the path forward to enabling the widespread use of lithium anodes.”
Balsara, a professor of chemical engineering at UC Berkeley, described this research in a paper in Nature Materials. This is the first study to employ microtomography using monochromatic beams of high energy or “hard” X-rays, which allows three-dimensional imaging of solid objects at a resolution of approximately one micron.
“We observed crystalline contaminants in the lithium anode that appeared at the base of every dendrite as a bright speck,” says Katherine Harry, lead author of the Nature Materials paper. “The lithium foils we used in this study contained a number of elements other than lithium with the most abundant being nitrogen. We can’t say definitively that these contaminants are responsible for dendrite nucleation, but we plan to address this issue by conducting in situ X-ray microtomography.”
Balsara and his group also plan further study of the role played by the electrolyte in dendrite growth, and they have begun to investigate ways to eliminate non-conductive impurities from lithium anodes.