Porous electrodes have been 3D printed in the past using extrusion processes in which a strand of material is extruded from a heated nozzle. Interdigitated, or clasped, structures created with this technique provide space for ion flow, but their limited geometries are quite inefficient. Carnegie Mellon University’s new research has developed a completely different additive process that allows for more complex geometries with increased ion flow potential.
The new technique takes advantage of an Aerosol Jet 3D printing system, and individually places droplets to form microlattice structures. Standard batteries do not utilize 30% to 50% of their electrode volume. The microlattices allow for higher ion flow rates and can increase electrode utilization. Specifically, the researchers created silver-based electrodes that showed a 4x increase in specific capacity and a 2x increase in areal capacity when compared to solid silver electrodes. These 3D printed batteries have higher specific energy than solid electrodes, so they can provide equivalent capacities with less material (and weight) if needed. The researchers project that this technology will be available for industrial applications in two to three years.
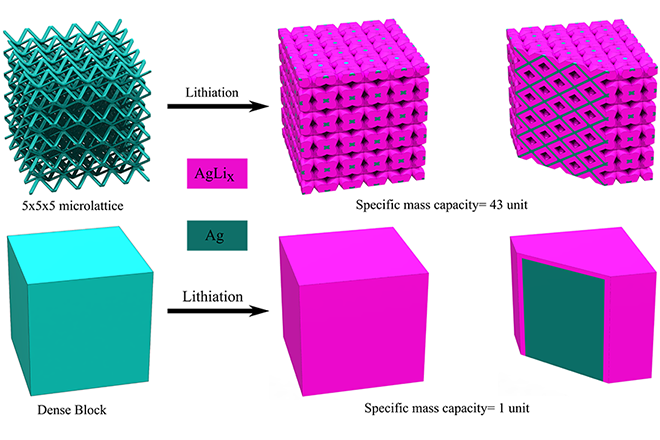 Lattice architecture can provide channels for effective transportation of electrolyte inside the volume of material, while for the cube electrode, most of the material will not be exposed to the electrolyte.
Lattice architecture can provide channels for effective transportation of electrolyte inside the volume of material, while for the cube electrode, most of the material will not be exposed to the electrolyte.
“In the case of lithium-ion batteries, the electrodes with porous architectures can lead to higher charge capacities,” says Carnegie Mellon Associate Professor Rahul Panat. “This is because such architectures allow the lithium to penetrate through the electrode volume, leading to very high electrode utilization and thereby higher energy storage capacity. In normal batteries, 30-50% of the total electrode volume is unused. Our method overcomes this issue by using 3D printing – we create a microlattice electrode architecture that allows the efficient transport of lithium through the entire electrode, which also increases the battery charging rates.”
When differentiating standard 3D printing techniques from the new method, Panat says, “Because these droplets are separated from each other, we can create these new complex geometries. If this was a single stream of material, as in the case of extrusion printing, we wouldn’t be able to make them. This is a new thing. I don’t believe anybody until now has used 3D printing to create these kinds of complex structures.”
Source: Carnegie Mellon University