The pulse of the industry: Wildcat Discovery Technology is at the forefront of battery research, solving problems and accumulating insight.
When Charged wants to catch up on the newest trends in cutting-edge battery technology, there are a few companies we turn to for insight. At the top of the list is a group whose high-speed research and discovery capabilities are so unique, that it helps other industry leaders solve their toughest problems.
Wildcat Discovery Technologies recently celebrated its 100th research project, working with over 60 companies since its inception in late 2006. The company was founded on the idea of adapting high-throughput combinatorial chemistry techniques – processes that are well known for discovering new pharmaceuticals – and using them to rapidly synthesize and evaluate energy-storage materials. The result is a very active research group that builds and tests prototype batteries up to 100 times faster than a standard lab.
Wildcat originally focused on hydrogen storage, but shifted to batteries in 2008. Since then the company has developed a list of battery materials for licensing or partnered development that reads like a syllabus for a graduate course in advanced battery technology (and that’s only the research projects it’s legally allowed to discuss with us).

New partners and new trends
Beyond developing materials internally, Wildcat works on many problems brought to them by battery companies from around the world. “Our collaboration model is taking off,” CEO Mark Gresser told Charged. “We’ve landed a bunch of new collaborative partners across different industries, including automotive OEMs, grid storage, and consumer electronics. Companies whose names you would know, but we can’t disclose right now.”
“In the early days, we did a lot of collaborations which were quite small,” he added. “But most of our projects now are much larger, and in many cases we’re involved in multi-year collaborations.”
The advanced battery industry has been on a roller coaster ride since 2008. There’s been an influx of government and venture funding, promising new markets beginning to find traction, some big failures, consolidations, and refocused efforts.

The folks at Wildcat say that they’ve noticed some changing trends as the industry has matured. “A few years back, people were really focused on the big energy discovery,” said Gresser. “Today, it feels like there is a more practical approach, and companies are focusing not just on energy, but power and all other metrics combined. There are still a lot of companies, including us, who are going for the big energy discoveries, but there’s also more work going on in a lot of different areas.”
As a handful of lithium-ion battery manufacturers are rising up as the top suppliers of high-energy EV batteries, others are looking for different niches to target. In the past year, for example, some companies have publicly stated their intent to focus on micro-hybrid solutions or stop-start batteries – a segment that is expected to grow significantly in the next five years.
Because of the varying demands of each application, this means a lot of incremental research and discovery work to try to make existing chemistries a little better for a particular application. While hybrid systems like stop-start technology aren’t quite as sexy as extended-range EVs, it’s a very viable technology to improve efficiency. Some predict that with slight improvements to batteries, it could be standard in all vehicles within a decade.
To reach these markets, Li-ion battery builders are “broadening their portfolio,” said Dee Strand, Chief Scientific Officer at Wildcat. “Instead of just high-energy materials, they’re looking at nearer-term solutions. Within each category, there is a lot of tailoring that occurs. Even if you look at a product like nickel-manganese-cobalt (NMC), there’s NMC at various compositions, run at various voltages. There are the gradient NMCs that claim to offer the best of both worlds. So, a lot of people are looking, even within a category, at tailoring a chemistry for exactly what they want in terms of power, energy and life.”

Making incremental improvements to existing battery technology requires a deeper understanding of what’s taking place in the cells, and more sophisticated research techniques. One of the interesting by-products of Wildcat’s collaboration model is its ability to identify gaps in the research capabilities of its many partners, and then create new experiments to improve areas of research that are tough for others to perform.
Gas detection
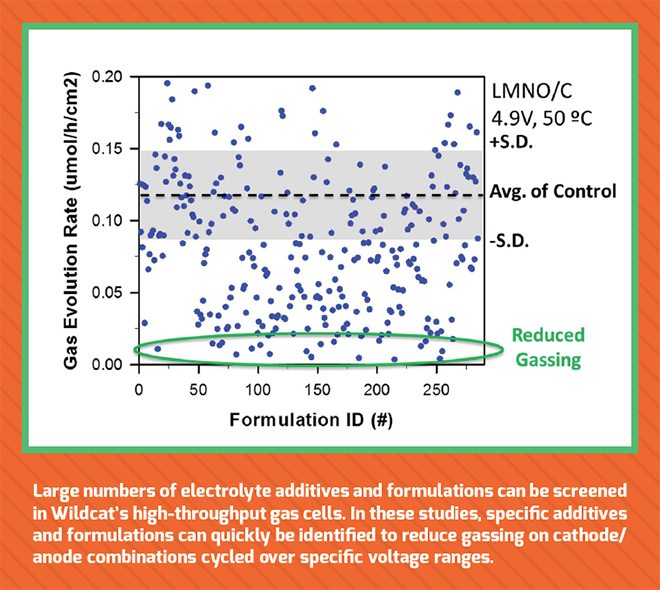
For instance, Wildcat touts its gas-sensing channels that can simultaneously measure gasses released by cells, along with the normal electrochemical measurements of capacity, coulombic efficiency and cycle life. Gas generation is a common problem for Li-ion batteries that leads to reduced cycle life and cell failure. It’s especially problematic as cell makers strive to enable cells to function at increased voltages in the quest for greater energy density.
Wildcat has developed high-throughput in situ gas measurement cells that allow for precise detection of gassing directly inside the cell. The new method enables continuous gas measurements while cells cycle. This unique approach, coupled with the company’s other techniques, lets its speedy scientists rapidly evaluate gas evolution for thousands of electrolytes in full cells, dramatically accelerating the development of improved additives and electrolyte formulations.
With such a tool, it’s possible to start discovering very interesting correlations. For example, if certain materials have poor coulombic efficiency, do they also have a lot of gas generation? Or if materials have poor high-temperature cycle life, is that accompanied by a particular gas being released?

“Lithium-titanate, for instance, is a chemistry that a lot of people like because it works well at low temperatures, and you don’t have to form a solid-electrolyte interface (SEI), but it tends to generate a lot of gas,” said Strand. “With our gas-sensing channels, we can test different variations to identify and discover solutions for gassing. We’ve had a lot of success in many different gas generation projects.”
Hot and cold
Wildcat has been involved in many projects to investigate electrolytes that work well in a wide temperature range. Measuring the performance of a test cell at high and low temperatures, in general, is something that everyone does. However, to do it efficiently is difficult, because it usually involves moving a lot of batteries to different environmental chambers held at various temperatures.
So, the company developed tools to make those measurements more automated. Essentially, it can independently control the temperature of each cell being tested across a wide temperature range (-40 degrees C to +70 degrees C), and importantly, vary cell temperatures rapidly without physically moving the cells.
“That’s really been a useful capability that allows us to move much quicker,” said Strand.
Electrode design
Another project that Wildcat has focused on is the effect of the electrode design on performance. “How does electrode loading or porosity affect the power, energy, and life?” asked Strand. “We can look at how ranging temperatures play into the electrode design. For example, if you have a porous electrode you might be limited by mass transport of your electrolyte at low temperatures. If it’s less porous, things can get more severe.”
Drive cycles
It would be nice if batteries in vehicles simply charged and discharged at a constant current, but that isn’t what happens in real life. On the road, the driving loads are a series of pulses. So, it’s important that new chemistries are tested using realistic drive-cycle protocols.
“We’re now testing cells running a lot of drive-cycle protocols,” said Strand. “The question is whether you will get the same performance when the cell is discharged using a drive-cycle protocol as a standard protocol. We’ve been working hard trying to understand those correlations over the past year.”
All of Wildcat’s expanded capabilities have been pushed by its partners and have required things like software rewrites and reengineered, or additional, equipment. The result is an expanding suite of discovery tools for the company to offer customers, but it also paints a larger picture of a battery industry that’s quickly evolving and continuing to gain traction and sophistication.
The talent pool
Another clear indicator of a hot market is a fight for talent. You may have seen some recent headlines about companies poaching each other’s battery experts (and the lawsuits that ensued). Throughout the world, battery scientists continue to be in high demand, and as Wildcat continues to expand, it must compete to attract the best of the best.
“We have a couple of pretty good selling points,” said Gresser. For one, the company’s located in San Diego where it’s perpetually 70-something degrees and sunny.
“Another huge advantage is our high-throughput work flow,” added Strand, “especially for those who are frustrated doing battery research using more traditional means, where you’re always fighting over channels and making compromises on the replicates you can do. Or, you have one reactor and it takes two or three days to make a material, so maybe you make one or two materials a week.”
What drives many into a life of science is the thrill of pushing technology to new heights – doing or discovering things for the first time in history. At times, however, the slow speeds on the road from theory to experiment to commercialization can be very frustrating.

“We tell recruits that when you get involved in a project here, you’ll do more experiments in six months then you’ve done in your entire career to date. And that’s very exciting,” said Gresser. “They love to come in every day, because they’re finding new things and they’re finding them fast. For a scientist it’s invigorating. It’s not just the number of experiments – it’s the diversity of the projects and problems they’re trying to solve. It’s never boring here, and that helps.”
A faster future
In many ways, following the progress of Wildcat is like taking the pulse of the energy storage industry. The company that started with its eye on developing new materials that could store hydrogen, quickly switched gears to focus on batteries, and is now rapidly expanding its capabilities.
Interestingly enough, Wildcat reports seeing something of a renewed interest in hydrogen research worldwide, and says that it may revisit that type of discovery if the uptick continues. “A lot of the tools that we originally developed to execute hydrogen research have carried over to our battery business,” said Gresser. “The way we synthesize materials – solid-state materials in particular – is very similar to the way we started out synthesizing complex metal hydrides. As well as some of the sealing technologies originally developed for hydrogen molecules. So, we may look at it again in the future, but right now we have our hands completely full with battery research.”
Wildcat continues to build momentum as it adds to its discovery techniques. In fact, it’s currently looking to increase the speed of its growth. The company is talking to a handful of its best customers about strategic funding to build some new improvements that are in the minds of its scientists and engineers. “We’re hoping to expand our capacity this year and to do it fairly rapidly to continue the growth trend,” said Gresser. “That will lead to more discoveries faster, and that’s always been our goal.”
High-speed science
Among Wildcat’s long list of discovery projects, here are a few recent ones that it can openly discuss.
Electrolytes
A lot of companies are taking more traditional materials, like NMC or lithium-cobalt oxide, to higher voltages. And, on many fronts, Wildcat has been working on electrolytes to enable long cycle life for those materials taken to higher voltage.
EM3 is the company’s latest modular design for electrolyte additives. It combines a core-forming molecule with a functional group that imparts some desirable property, like high-voltage stability. EM3 has been demonstrated to have a significant effect in improving the cycle life of traditional cathode materials operated at higher voltages and/or higher temperatures.
Silicon anode electrolytes
The company has also been working on electrolytes that improve the performance of certain high-energy anode materials. One DOE-funded project aims to develop electrolytes and electrolyte additives that perform well with silicon anodes.
Silicon anodes enable substantial improvements in energy density and cost, relative to current Li-ion tech. However, they have a very large volumetric change when cycling, so the SEI layer that forms often cracks. Then it forms more SEI and ends up with a very thick resistive film on the anode, which causes it to lose both capacity and power.
Wildcat says that its new electrolyte project, EM4, has been very successful at making more mechanically robust SEI layers on the anode. So, as volume changes occur, the SEI doesn’t crack.
Lower energy, big potential
On the lower-energy front, which is just as important for the auto industry, there’s a lot of effort towards micro-hybrids. These batteries need to have good power and cycle-life performance over a wide temperature range. So, Wildcat has recently been working on electrolyte formulations that enable wide temperature ranges.
“It’s really hard to find electrolytes that give you low resistance at low temperature,” said Strand. “You don’t want them to become too viscous, or have too low a conductivity, or make too resistive of an SEI layer, and at the same time they need to have high temperature stability. A lot of the chemistries where the approach is to reduce the impedance at low temperatures generally cause the materials to be less stable at high temperatures. So, we recently had some discoveries for an additive that provide benefits at both low and high temperatures, which is really important to the automotive industry.”
Solid-state
Another area that Wildcat is engaged in is solid-state electrolytes (SSE). SSE technology has a number of advantages. It can improve energy density in large-format cells by using a lithium anode without the worry of dendrites, because it has a solid on top of it. Dendrites are growths that can form on the surfaces of anodes during cycling, reducing battery life or even causing a catastrophic short circuit. SSE can also reduce packaging requirements, because there is no worry of liquid electrolyte leaking out or gasses being created.
Wildcat’s effort in SSE involves making very practical and processable films. The company says that some of the ceramics options are very brittle and hard to process. “We want to make a processable film with high ionic conductivity,” said Strand. “Our approach has been to use composites of ceramics with a polymer to try to get the best of both worlds – the advantages of the ceramics and the processability of the polymer.”
Helping others, helping themselves
Everybody wants to bring down costs, particularly the cost of cathode materials.
Conveniently enough, one of the things that Wildcat’s high-throughput process has demonstrated to be great at is finding the process variables that can make the best-performing material at the lowest cost.
The techniques can identify important variables like the energy intensity of a process, the time it takes to complete different unit operations, the utilization of capital investment on things like expensive furnaces, etc.
Wildcat has begun working with material suppliers to bring down their production costs of existing cathode materials. These customers aren’t asking Wildcat to discover a new cathode composition, but to work with their preferred composition and help them figure out how to improve scale, performance, and cost.
“And it turns out we can do that really well,” said Gresser. “With our system, we’re able to run experiments to look at particle size, particle size distribution, morphology, etc. All the different things that are used as processing additives or in the process, like types of solvents. Basically, it’s a huge amount of experiments and very complex, like all of our research. In the end, you get a refined process for scaling these materials and with really good battery performance.”
This article originally appeared in Charged Issue 18 – March/April 2015.