Still looking for a replacement for lithium? How about magnesium? It’s divalent, so it can displace double the charge per ion (i.e., Mg2+ rather than Li+). It’s much more abundant than lithium, and more stable. However, researchers investigating Mg-ion batteries have run into several limitations, including anode/electrolyte incompatibility.
Now a team at the Toyota Research Institute of North America (TRINA) has reported a major advance in the the development of electrolytes for magnesium (Mg) batteries.
In “An Efficient Halogen-Free Electrolyte for Use in Rechargeable Magnesium Batteries,” published in the journal Angewandte Chemie, the researchers, led by TRINA’s Dr. Rana Mohtadi, describe how they developed an electrolyte based on a magnesium monocarborane salt (MMC) that is compatible with Mg metal, has high anodic stability, and is non-corrosive.
The development of a non-corrosive electrolyte enabled the first demonstration of a high-voltage coin cell battery. “This achievement is a turning point in the research and development of Mg electrolytes that has deep implications for realizing practical rechargeable Mg batteries,” wrote the scientists.
“Mg metal offers high volumetric capacity (3,833 mAh cm3 vs. 2,036 mAh cm3 for Li metal) while being non-dendritic and abundant in the Earth’s crust. Since Aurbach et al demonstrated the first rechargeable Mg battery prototype, challenges toward realizing Mg batteries still remain. These stem from the absence of practical electrolytes and high capacity/high voltage cathodes. The field demands electrolytes capable of operating at high voltages whilst being compatible with Mg metal and all other battery components.”
In 2014, Mohtadi and her TRINA colleagues demonstrated the effectiveness of Mg(BH4)2 as an electrolyte – the first non-organomagnesium electrolyte compatible with Mg metal. Since then, the team has been seeking materials with high oxidative stability.
“High oxidative stability is crucial for the development of Mg batteries for operation with future high-voltage cathodes. Although organomagnesium compounds with oxidative stability above 3 V (vs Mg) have been reported, most have significantly reduced oxidation onset potentials when deposition and stripping occurs on non-noble-metal surfaces. Mg(BH4)2 displayed enhanced stability on these metals. We therefore sought to identify an electrolyte candidate which would retain the attractive properties of Mg(BH4)2 but offer enhanced oxidative stability.”
Pursuing a new design concept involving boron cluster anions, the team found that monocarborane CB11H12- produced the first halogen-free, simple-type Mg salt that is compatible with Mg metal and displays an oxidative stability surpassing that of ether solvents.
Source: Angewandte Chemie via Green Car Congress
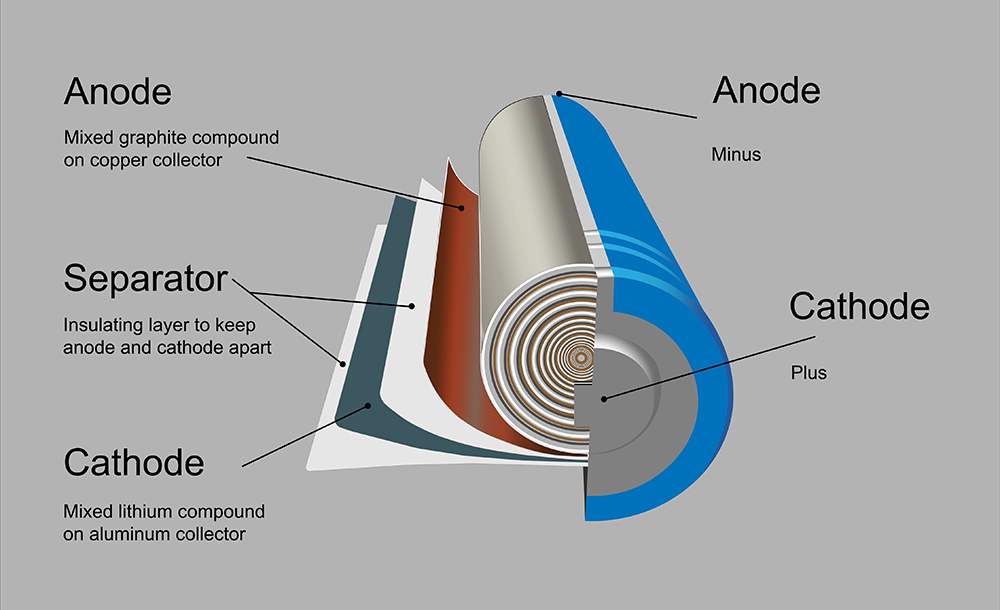
Image: O. Usher (UCL MAPS) – (CC BY 2.0)