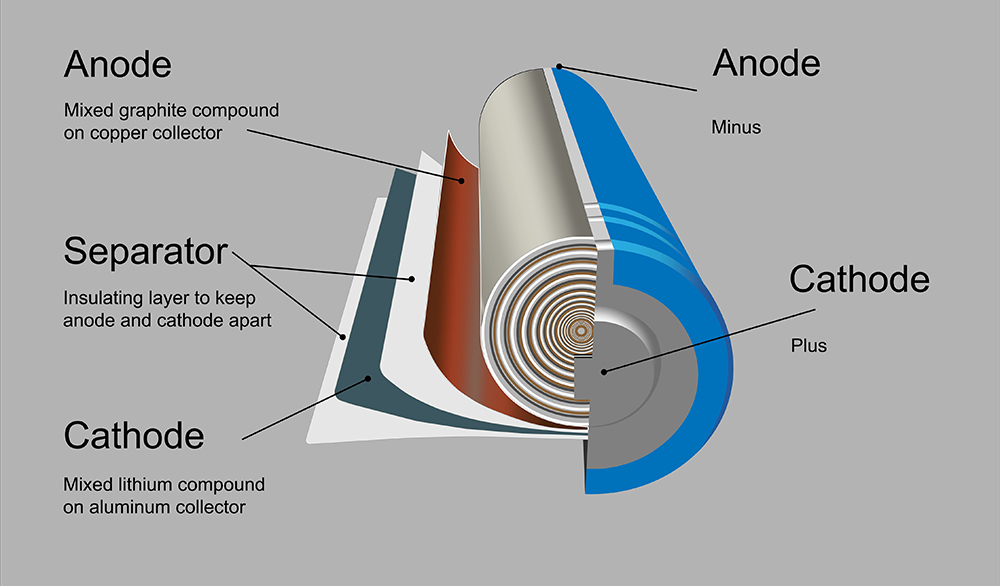
Researchers at the University of Maryland have developed a new template that they hope will enable a safer alternative to liquid electrolytes, which have several drawbacks, including flammability, leakage and dendrite formation in the electrodes.
In “Lithium-ion conductive ceramic textile: A new architecture for flexible solid-state lithium metal batteries,” published in Materials Today, UMD researchers Eric Wachsman, Liangbing Hu and colleagues explain how they developed a solid-state battery using a non-flammable ceramic electrolyte known as garnet.
The research team used available fabrics as a template for creating lithium-conducting garnet fiber mat textiles, then filled the pore space between the fibers with a solid polymer electrolyte. The result was a scalable process to create hybrid ceramic/polymer lithium electrolytes with higher conductivity and strength.
“The goal of this research is to develop safe solid-state components for use in lithium-metal batteries,” said lead author Dr. Yunhui Gong. “Such components should have good ionic conductivity, abundant surface area and scalable production potential. We have successfully developed a garnet-fiber textile to meet these key requirements.”
“Nothing like this has ever been done before,” said Dr. Wachsman. “The resulting hybrid structure is capable of fast-ion conduction through the continuous ceramic fibers, but with the flexibility of more traditional polymer electrolytes. What’s more, the garnet fibers will help block the formation of Li-dendrites, thus enabling higher capacity Li-metal anodes.”
The research group will continue its research efforts with the goal of making the textile thinner to reduce resistance to ionic transport between electrodes.
Source: University of Maryland