Sponsored by OCSiAl Group
Li-ion batteries represent the most dynamic and fastest growing battery market, with total sales of $10 billion in 2015 [i]. Historically, the primary challenge for battery manufacturers has been the constant demand for greater energy density, both volumetric and gravimetric. Li-ion’s dramatic rise is in large part due to solving this challenge more successfully than other battery technologies.
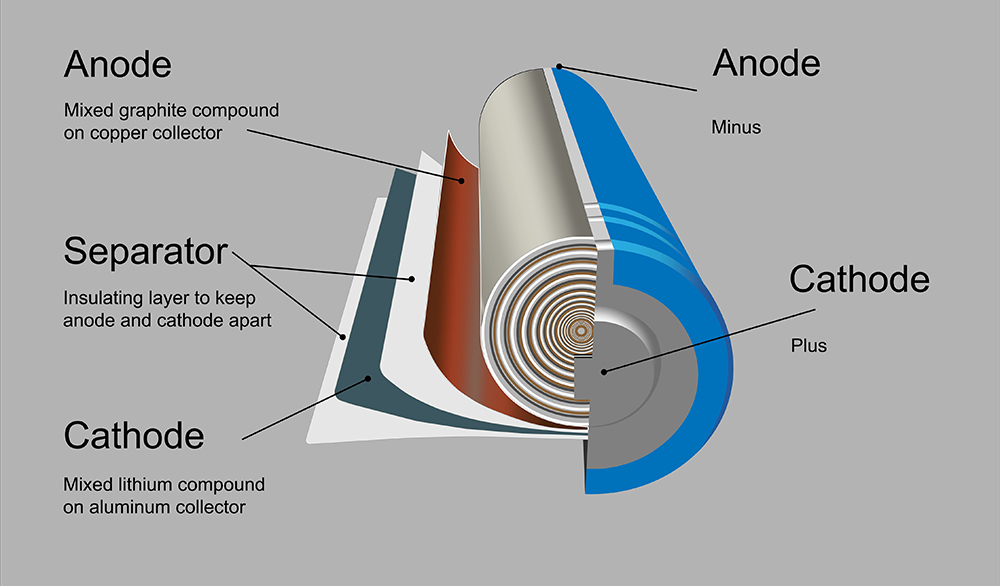
Among other improvements, the amount of inactive material in battery electrodes has steadily been reduced through the development of better binders and conductive additives. Over time, high-performance conductive carbon black and multi wall carbon nanotubes (MWCNTs) have replaced regular conductive carbon black and graphite, enabling electrode formulations with as much as 96–98% active material. Further reductions in inactive material are giving companies an edge in this highly competitive market.
Recently, single wall carbon nanotubes (SWCNTs) have been gaining ground in the industry as they provide superior conductivity at ultra-low concentrations, allowing the amount of active material in a battery electrode to be increased up to 99.5%. This article reviews the ways SWCNTs are helping solve energy density challenges in commercial li-ion battery formulations today. Additionally, future applications in lithium-ion and lead acid batteries are discussed.
TRENDS TODAY: COMMERCIALLY AVAILABLE SWCNT SOLUTIONS
SWCNTs replace conductive carbon black, graphene and MWCNTs in battery electrodes
In comparison with traditionally used conductive additives such as conductive carbon black or recent additives including MWCNTs and graphene, SWCNTs enable conductivity starting from a concentration of just 0.001%. Low-rate systems, such as batteries for cell phones and laptops based on lithium cobalt oxide (LCO) chemistry, have been shown to work well with 0.02–0.06% of SWCNTs.
Recent trials in high-rate automotive batteries conducted by Aleees also showed that a significant reduction in the amount of binder and a remarkable 10% increase in energy density can be achieved just by replacing the conductive carbon black and graphite in LFP cathodes with 0.1% of SWCNTs.
Replacement of “old” conductive additives with SWCNTs is already occurring for a wide range of manufacturers as the novel additive completes testing programs across the globe. Mass production of lithium-ion batteries containing SWCNTs in the cathode is estimated to start in early 2018 with additional manufacturers starting production throughout 2018.
Si-C, Si-O SWCNT structures for high energy density batteries
The use of nanotubes is not just limited to replacing other conductive additives. At an international seminar held in June 2016 in Shenzhen that was devoted to the use of SWCNTs in electrochemical power sources, BAK, a large manufacturer of Li-ion batteries in China, shared their results and commercial plans using SWCNTs in silicon anodes (Si-C).
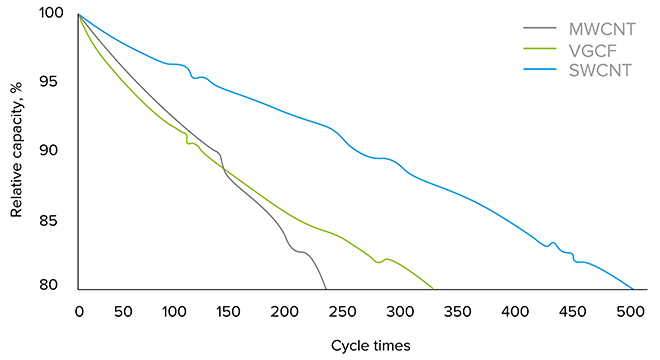 Figure 1: SWCNT comparison in Si-anode high-energy 18650 cells
Figure 1: SWCNT comparison in Si-anode high-energy 18650 cells
The cycle life of the 3.5 Ah 18650 cells with Si-C was increased from 350 to 500 cycles when a small amount of nanotubes was added to the anode formulation in the form of TUBALL BATT H2O, a water-borne dispersion of SWCNTs sold by OCSiAl (Figure 1).
FUTURE TRENDS: SWCNT–BASED SOLUTIONS UNDER DEVELOPMENT NOW
Silicon anodes and SWCNT structures for future high energy density batteries
Lab studies by OCSiAl have shown that a new composite material made of silicon layers deposited on SWCNTs in sandwich-like structures can achieve remarkably high capacity, over 2500 mAh/g, and a long cycle life (Figures 2 and 3)[ii].
The challenge with silicon materials is that their volume changes up to 400% during charge and discharge, which leads to rapid degradation of the electrode structure and a short cycle life. It was observed that the SWCNT layers in this sandwich successfully accommodate volume changes during lithium intercalation into silicon and prevent degradation of the material. Si-SWCNT composites using this technique could decrease battery weight by 20% and size by 25%.
 Figure 2: SEM of silicon deposited on SWCNTs
Figure 2: SEM of silicon deposited on SWCNTs
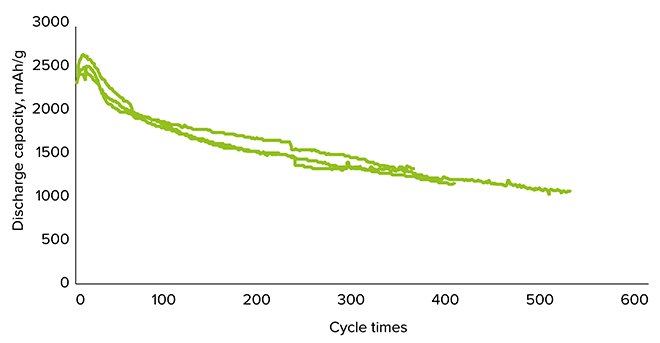 Figure 3: Cycle life in Si-SWCNT composite sells
Figure 3: Cycle life in Si-SWCNT composite sells
Promising future for lead-acids with SWCNTs
The lead–acid battery is the workhorse of the battery world. With all the excitement about Li-ion cells, it’s easy to forget that manufacturing volumes of lead-acid batteries are about five times higher than that of Li-ions in terms of megawatt-hours, and they can be found in virtually every car, as well as in UPS and backup systems, in forklifts, and much more. They are cheap and reliable and work in the harshest conditions in both hot and cold climates. Interestingly, even Tesla’s electric vehicles have 12V lead-acid batteries that power a host of 12V electronics.
Lead-acid batteries were invented in the 1800s and there haven’t been any significant changes in their chemistry since that time. Recently, they have been facing competition from Li-ion cells and performance challenges in many applications, notably telecom and automotive start-stop systems, where faster charging and the ability to operate in a low state of charge over many cycles are required.
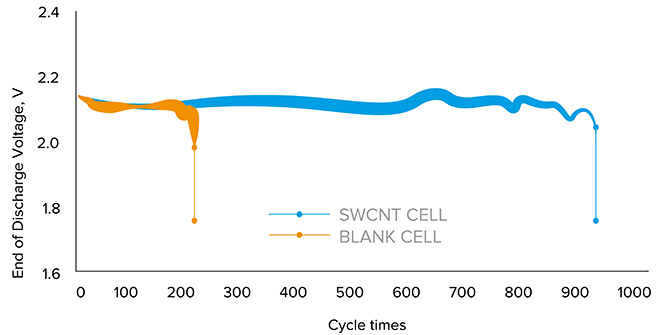 Figure 4: Lead-acid batteries achieved a four-fold increase in cycle life with just 0.001% SWCNT
Figure 4: Lead-acid batteries achieved a four-fold increase in cycle life with just 0.001% SWCNT
Recently several research groups have initiated early studies on the potential of SWCNT in lead-acid batteries.
Presenting at the world’s first Nanoaugmented Materials Industry Summit in November 2016, Professor Doron Auerbach, the head of the chemistry department at Bar-Ilan University, said there had also been remarkable results with the addition of SWCNTs to the electrodes of advanced lead batteries with gel electrolytes.
“It was above all expectations,” said Auerbach. “Single wall carbon nanotubes increased the mechanical strength of the active mass, the integrity of the active mass and the electrical conductivity of the active mass. Even tiny amounts were very effective, which means that with mass production the cost will not be a big issue because the amounts required are so tiny.”
Pioneering studies showed that with just 0.001% of SWCNTs added to the electrode paste of ordinary lead-acid batteries, there was a more than four-fold increase in cycle life while the rate capability also increased over four times.
“In advanced lead-acid batteries the electrolyte is now a gel, and we didn’t expect there to be much of a difference because the batteries are better, after all. But when the cells were polarized, they needed much lower voltage if they had SWCNTs added to the gel. When we added our magic additive we could see the improvement in advanced lead batteries, which was spectacular when compared with blank cells. There was a four-fold gain in cycle number,” commented the professor.
“What is more, we also discovered that the application of single wall carbon nanotubes allows us to suppress sulfation,” added Aurbach.
These impressive results are blazing a trail for future studies and developing TUBALL nanotube-based off-the-shelf technologies for lead-acid battery applications.
Commercial availability
While SWCNTs have long been out of reach for commercial customers due to lack of availability and price, this issue has largely been solved. OCSiAl Group, the largest manufacturer of SWCNTs, had an annual capacity of 10 tonnes in 2016, or about 90% of global production, enough to supply SWCNTs for nearly every high-performance Li-ion application on the market.
The technology for introducing SWCNTs into manufacturing processes is also just as important as the wide availability of nanotubes. Today, more and more companies are beginning to make dispersions of SWCNT in water and in NMP, the two solvents most often used in battery manufacturing. Sold under the name TUBALL™ BATT, OCSiAl has developed several ready-to-use SWCNT concentrates for the battery industry along with key Asia Pacific suppliers Kusumoto Chemicals and Haiyi. Industry giants such as LANXESS are also using TUBALL nanotubes to produce high performance conductive materials using their own recipes and technologies.
For more information contact usa@ocsial.com or visit them at The Battery Show booth #1602, held September 12-14, 2017 in Novi, Michigan, USA.
References:
[i] Energy Storage, Frost & Sullivan (2009)
[ii] O. Bobrenok et al, High Capacity Hybrid Si-SWCNT Anode Structures for Li-Ion Batteries, 228th ECS Meeting, Phoenix, AZ, October 11–16, 2015