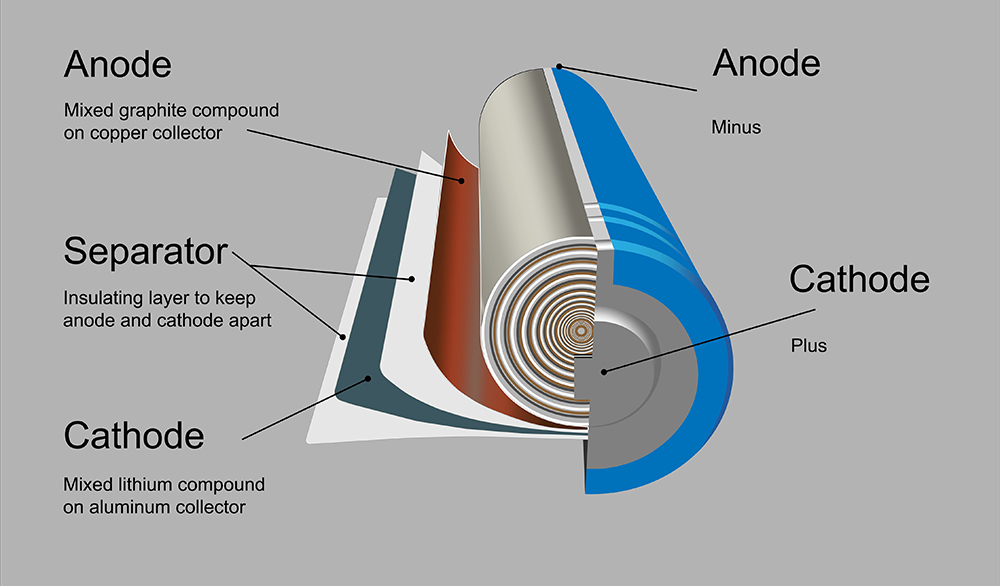
Current electrode materials such as graphite are able to stably adsorb only a limited number of lithium ions, an inconvenient truth that restricts capacity. That’s why researchers around the world are investigating semiconductor materials such as silicon as alternative electrodes.
Bulk silicon is able to accommodate enormous quantities of lithium, but the migration of the lithium ions destroys the silicon’s crystal structure. Now a team from the Helmholtz-Zentrum Berlin (HZB) Institute of Soft Matter and Functional Materials has observed for the first time how lithium ions migrate into thin films of silicon.
In “Lithiation of Crystalline Silicon As Analyzed by Operando Neutron Reflectivity,” published in the journal ACSnano, Professor Matthias Ballauff and colleagues show that extremely thin layers of silicon may be sufficient to achieve the maximal load of lithium.
The team was able to observe a lithium-silicon half-cell during its charging and discharge cycles, and found that lithium ions do not penetrate deeply into the silicon. During the charge cycle, a 20-nanometer anode layer develops, containing a high proportion of lithium.
SEE ALSO: Tesla tweaks its battery chemistry: a closer look at silicon anode development
“We were able to precisely track where the lithium ions adsorb in the silicon electrode using neutron reflectometry methods, and also how fast they were moving,” says Dr. Beatrix-Kamelia Seidlhofer, who carried out the experiments using the neutron source at the Institut Laue-Langevin in Grenoble, France.
Dr. Seidlhofer discovered two different zones. Near the boundary with the electrolytes, a roughly 20 nm layer formed, with extremely high lithium content: 25 lithium atoms were lodged among 10 silicon atoms. An adjacent layer contained only one lithium atom for ten silicon atoms. Both layers together are less than 100 nm thick after the second charging cycle.
After discharge, about one lithium ion per silicon node in the electrode remained in the silicon boundary layer exposed to the electrolytes. Seidlhofer calculates from this that the theoretical maximum capacity of these types of silicon-lithium batteries is about 2300 mAh/g. This is more than six times the theoretical maximum capacity for a lithium-ion battery constructed with graphite (372 mAh/g).
Source: Helmholtz Center Berlin