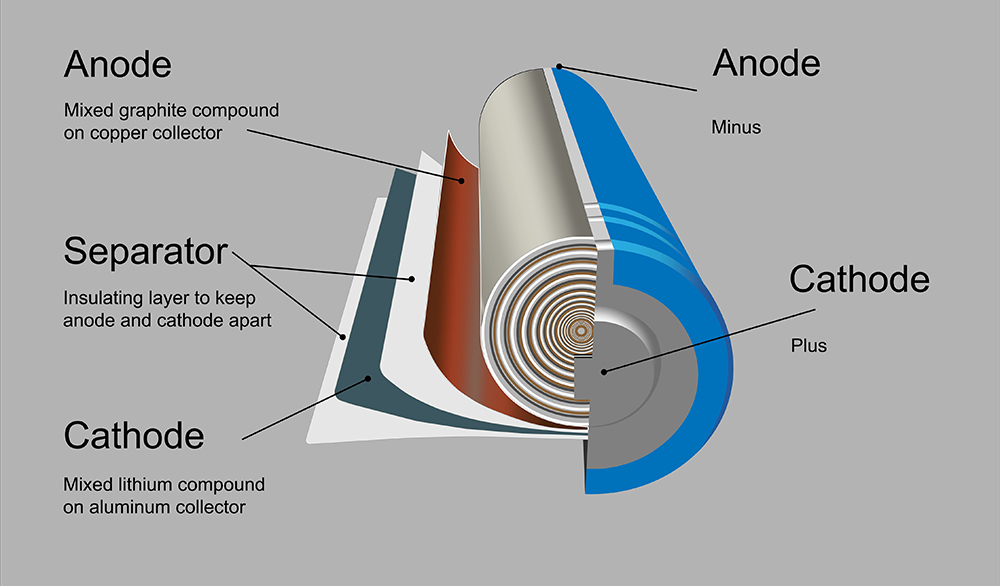
Michigan Technological University researchers exhort you to take pity on “the poor lithium ion.” As it endlessly rushes from anode to cathode and back again, all kinds of chemical processes take place, eventually causing batteries to lose capacity, and possibly even overheat.
Much of what goes on at the atomic scale as lithium-ion batteries are charged and discharged is still poorly understood, and scientists around the world are using a variety of imaging techniques to get a better look at the process.
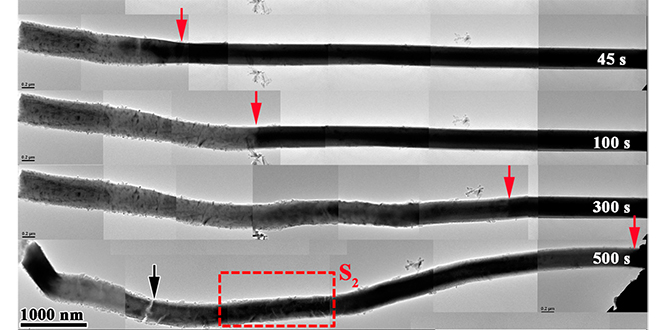
A research team led by Reza Shahbazian-Yassar, a Professor in Nanotechnology at Michigan Tech, has documented what can happen to anodes as lithium ions work their way into them, using transmission electron microscopy. The paper, Lithiation-Induced Shuffling of Atomic Stacks, was published in the journal Nano Letters.
“We call it atomic shuffling,” says Shahbazian-Yassar. “The layered structure of the electrode changes as the lithium goes inside, creating a sandwich structure – there is lots of localized expansion and contraction in the electrode crystals, which helps the lithium blaze a trail through the electrode.”
SEE ALSO: Could Sakti3 build GM’s 200-mile battery?
The atomic shuffling helps explain how lithium ions move through the anode, and provides a clue as to why most anodes made of layered materials eventually fail. “We showed that the ions cause a lot of local stress and phase transitions,” said team member Anmin Nie.
Source: Michigan Technological University
Image: Shahbazian-Yassar et al.