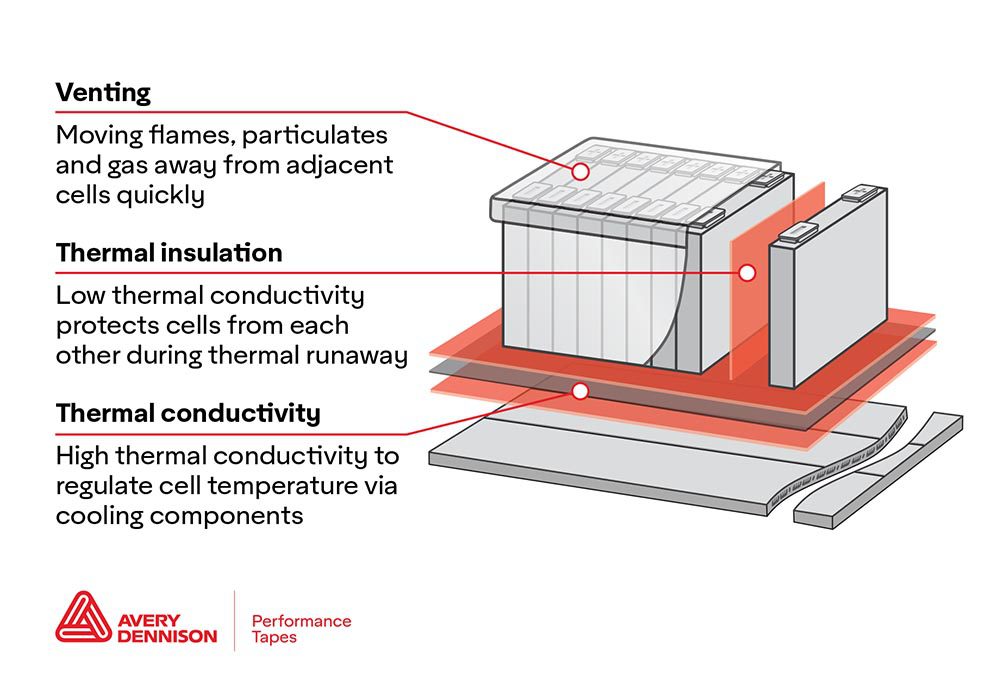
A team of researchers affiliated with the South Korean university UNIST has developed a new way to increase the energy efficiency of metal-air batteries by using a conducting polymer.
In “Polypyrrole-assisted oxygen electrocatalysis on perovskite oxides,” published in Energy & Environmental Science, Professors Dong-Gyu Lee, Hyun-Kon Song and Guntae Kim explain that, in the cathode of a metal-air battery or fuel cell, oxygen is reduced to metal oxide or water. Catalysts are required to accelerate the reaction. Platinum is a common catalyst, but its high price is an obstacle to commercialization of the technology.
The researchers reported that the catalytic activity of perovskite, which can be substituted for platinum, was dramatically enhanced by adding a conducting polymer called polypyrrole. When the perovskite or polypyrrole is used alone, its activity is not equal to that of platinum. However, when the two are physically mixed, the catalytic activity was dramatically enhanced, matching that of platinum.
“The reaction by which oxygen receives electrons is oxygen reduction. The property of polypyrrole which is sensitive to oxygen [helps] to accelerate this reaction,” says Professor Song.
“Because the oxide-polypyrrole complex is made by a simple operation, the catalyst will be proper to apply to next-generation energy devices,” says Professor Lee.
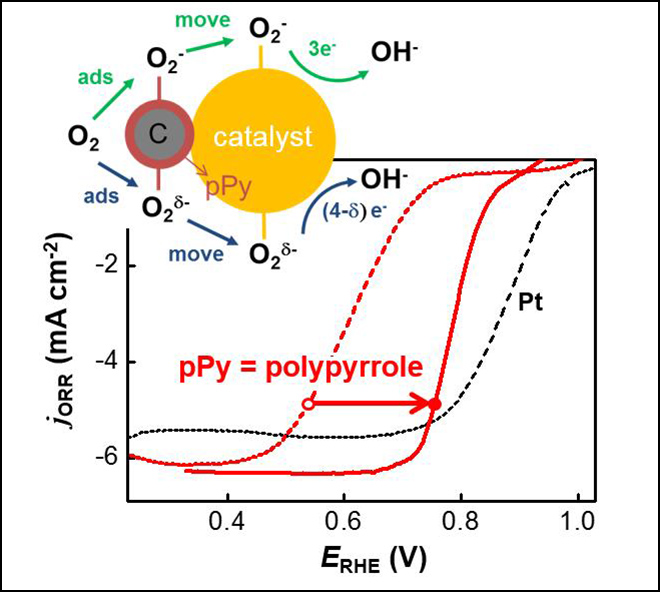 The oxygen reduction reaction at the anode of the cell is divided into four stages. The first step, which is the slowest, involves the addition of polypyrol to improve the catalytic reaction.
The oxygen reduction reaction at the anode of the cell is divided into four stages. The first step, which is the slowest, involves the addition of polypyrol to improve the catalytic reaction.
Source: UNIST