Using a new method to track the electrochemical reactions in a battery, scientists at the DOE’s Brookhaven National Laboratory have gained new insight into why fast charging inhibits performance. The results, published in Nature Communications, could help battery makers to optimize materials for faster-charging batteries.
“Our work was focused on developing a method to track structural and electrochemical changes at the nanoscale as the battery material was charging,” said Brookhaven physicist Jun Wang, who led the research. Her group was particularly interested in chemically mapping what happens in a lithium iron phosphate cathode as the battery charged. “We wanted to catch and monitor the phase transformation that takes place in the cathode as lithium ions move from the cathode to the anode.”
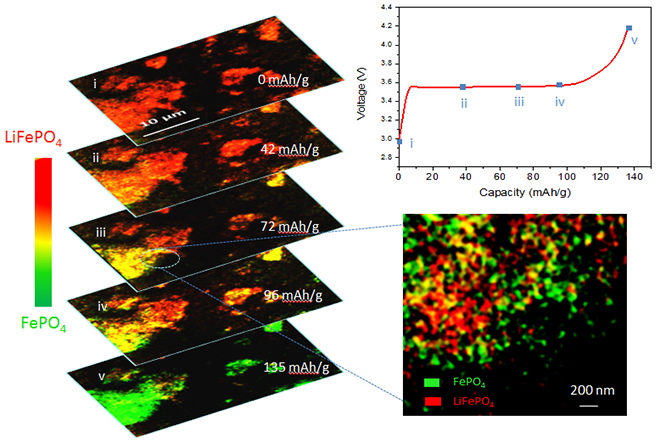
Getting as many lithium ions as possible to move from cathode to anode through this process, known as delithiation, is the key to recharging a battery to its fullest capacity. Understanding the subtle details of why that doesn’t always happen could lead to ways to improve battery performance.
The Brookhaven team used a combination of full-field, nanoscale-resolution transmission x-ray microscopy (TXM) and x-ray absorption near-edge spectroscopy (XANES). These x-rays produce both high-resolution images and spectroscopic data, revealing where lithium ions remain in the material, and where they’ve been removed, leaving only iron phosphate.
The scientists studied the delithiation reaction under two different charging scenarios -rapid and slow. At the fast charging rate, the images show that the transformation from lithiated to delithiated iron phosphate proceeds inhomogeneously. In some regions of the electrode, all the lithium ions are removed, leaving only iron phosphate behind, while particles in other areas show no change at all, retaining their lithium ions. Even in the “fully charged” state, some particles retain lithium, and the electrode’s capacity is well below the maximum level.
“This is the first time anyone has been able to see that delithiation was happening differently at different spatial locations on an electrode under rapid charging conditions,” Jun Wang said.
Slower charging, in contrast, results in homogeneous delithiation – lithium iron phosphate particles throughout the electrode gradually change over to pure iron phosphate – and the electrode has a higher capacity.
SEE ALSO: Phinergy CEO on aluminum-air batteries and 1,000-mile EVs
“People don’t want to charge slowly. We want to know why fast charging gives lower capacity,” said Jiajun Wang, the lead author of the paper. “Our results offer clues to explain why, and could give industry guidance to help them develop a future fast-charge/high-capacity battery.”
For example, the phase transformation may happen more efficiently in some parts of the electrode than others due to inconsistencies in the physical structure or composition of the electrode. “So rather than focusing only on the battery materials’ individual features, manufacturers might want to look at ways to prepare the electrode so that all parts of it are the same, so all particles can be involved in the reaction instead of just some,” Wang said.
Source: Nature Communications, Brookhaven National Laboratory via Green Car Congress