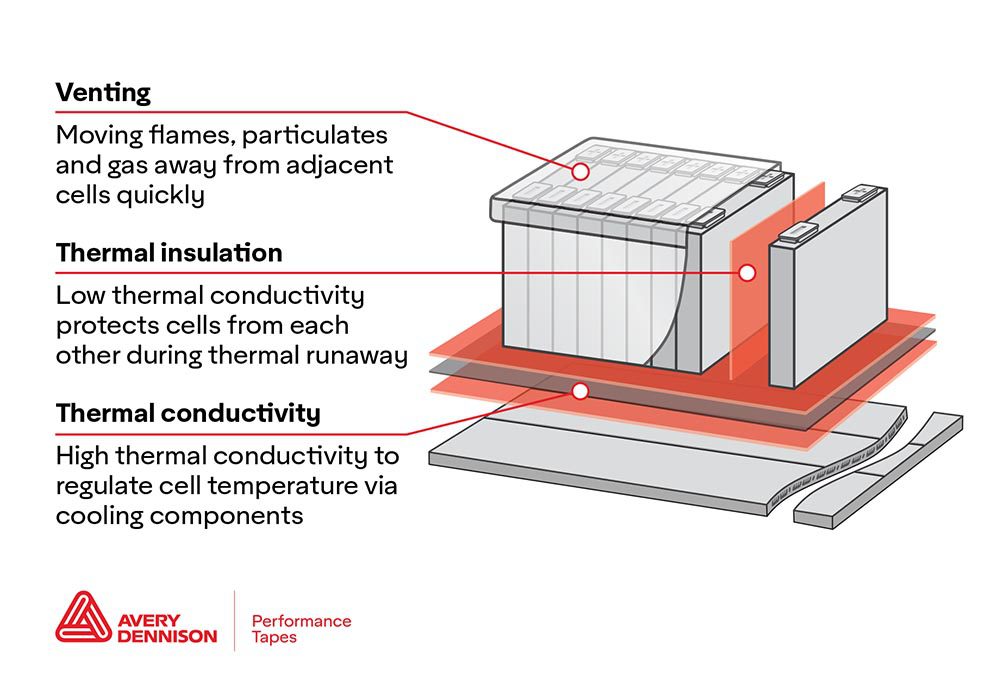
Researchers from the College of Chemistry at China’s Nankai University have announced a battery breakthrough using a new type of electrolyte.
In “Hydrofluorocarbon electrolytes for energy-dense and low-temperature batteries,” published in the journal Nature, the team explains how they designed and synthesized a series of new fluorinated hydrocarbon solvent molecules with fluorine coordination. Based on this, they constructed an electrolyte system that replaced the traditional lithium-oxygen coordination in electrolytes, enabling a 700 Wh/kg lithium metal battery to achieve reversible cycling.
Oxygen atoms have long been regarded as indispensable elements in the solvents of electrolytes. “Current lithium-ion battery electrolytes are usually composed of lithium salts and carbonate solvents,” the researchers explain. “The ion-dipole interaction between lithium and oxygen in the carbonate solvents can promote the dissolution of lithium salts. However, this solvent has poor wettability and requires a large amount, which makes it difficult to further increase the energy density of the battery. The strong interaction also hinders the interfacial charge transfer in the battery and limits low-temperature performance.”
The Nankai team tested the new electrolytes in lithium metal pouch cells, and were able to achieve specific energy exceeding 700 Wh/kg at room temperature and ~400 Wh/kg at -50° C. These hydrofluorocarbon (HFC) electrolytes thus offer a scalable solution for batteries operating in extreme cold.
Source: Nankai University