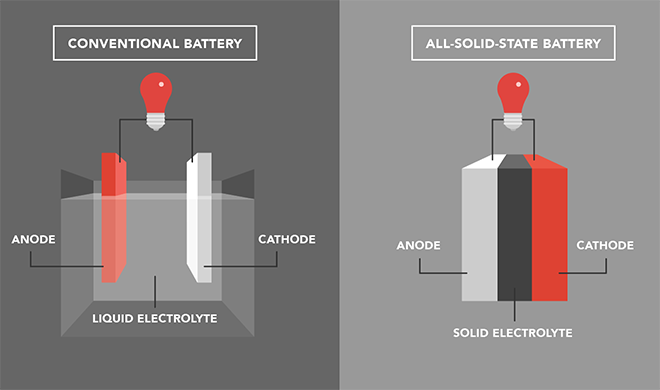
The race is on to develop batteries that are less expensive, safer, longer-lasting, more energy-dense, and easily recyclable. All-solid-state batteries show promise, but obstacles to their wide-scale adoption remain. In a review article published in Nature Nanotechnology, nanoengineers at the University of California San Diego (UCSD) offer a research roadmap that discusses four challenges of commercializing this technology:
1) Stable solid electrolyte chemical interfaces
2) New tools for in-operando diagnosis and characterization
3) Scalable and cost-effective manufacturability
4) Batteries designed for recyclability
“It’s critical that we step back and think about how to address these challenges simultaneously, because they are all interrelated,” said Shirley Meng, a nanoengineering professor at UCSD.
The researchers focused on inorganic solid electrolytes such as ceramic oxides or sulfide glasses. Inorganic solid electrolytes are a relatively new class of solid electrolytes for all-solid-state batteries (in contrast to organic solid electrolytes, which have been more extensively researched.) “At this point, we should shift our focus away from chasing higher ionic conductivity,” said Meng. “Instead, we should focus on stability between solid-state electrolytes and electrodes.”
The process of understanding what goes on inside a battery requires real-time nanoscale characterization. “We have a much easier time observing today’s lithium-ion batteries. But in all-solid-state batteries, everything is solid or buried. If you try the same techniques for all-solid-state batteries, it’s like trying to see through a brick wall,” said Darren Tan, a nanoengineering Ph.D. candidate at UCSD. One way researchers are overcoming these challenges is using cryogenic methods to keep battery materials cool, mitigating their decomposition under the electron microscope probe.
To overcome scalability issues, researchers are combining ceramics used in traditional material sciences with polymers used in organic chemistry to develop flexible and stable solid electrolytes that are compatible with scalable manufacturing processes. To address problems of material synthesis, the team also reports how solid electrolyte materials can be scalably produced using single-step fabrication without the need for additional annealing steps.
Today’s battery recycling methods are expensive, energy- and time-intensive, and include toxic chemicals for processing. Moreover, these methods only recover a small fraction of the battery materials, mainly because today’s batteries have not been designed with recyclability in mind. “Cost-effective reusability and recyclability must be baked into the future advances that are needed to develop all-solid-state batteries that provide high energy densities,” said UCSD nanoengineering professor Zheng Chen. “It’s critical that we don’t make the same recyclability mistakes that were made with lithium-ion batteries.”
Source: UCSD