In the quest to increase the energy density of Li-ion batteries, Yuan Yang, an Assistant Professor at Columbia Engineering, has developed a novel approach: reducing or eliminating capacity loss that occurs when a battery is first manufactured. In a paper recently published in Nano Letters, Yang and colleagues explain how this new method could improve Li-ion energy density by 10-30%,
“When lithium batteries are charged the first time, they lose anywhere from 5-20% energy in that first cycle,” says Yang. “Through our design, we’ve been able to gain back this loss, and we think our method has great potential to increase the operation time of batteries.”
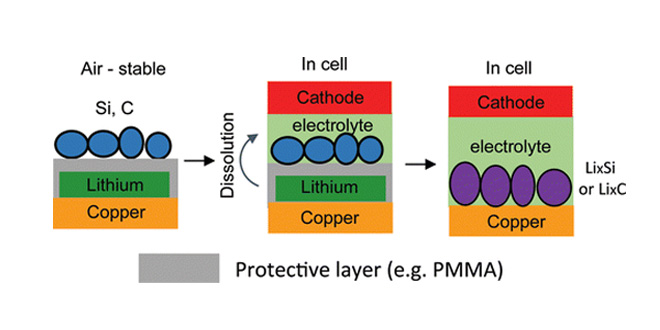
When a lithium battery receives its first charge after production, a portion of liquid electrolyte is reduced to a solid phase and coated onto the negative electrode of the battery. This process, usually done before batteries are shipped from a factory, is irreversible, and lowers the energy stored in the battery by as much as 30%.
The traditional approach to compensating for this loss has been to put certain lithium-rich materials in the electrode. However, most of these materials are not stable in ambient air. Manufacturing batteries in moisture-free air is much more expensive than manufacturing in ambient air. Yang has developed a new trilayer electrode structure to fabricate lithiated battery anodes in ambient air.
In these electrodes, he protected the lithium with a layer of the polymer PMMA, coated the PMMA with active materials such as artificial graphite or silicon nanoparticles, then dissolved the PMMA layer in the battery electrolyte, exposing the lithium to the electrode materials. “This way we were able to avoid any contact with air between unstable lithium and a lithiated electrode,” Yang explains, “so the trilayer-structured electrode can be operated in ambient air.”
“This three-layer electrode structure is indeed a smart design that enables processing of lithium-metal-containing electrodes under ambient conditions,” notes Hailiang Wang, Assistant Professor of Chemistry at Yale University, who was not involved with the study. “The initial Coulombic efficiency of electrodes is a big concern for the Li-ion battery industry, and this effective and easy-to-use technique of compensating for irreversible Li-ion loss will attract interest.”
Source: EurekAlert!