A team of researchers has identified a new class of non-flammable electrolytes for lithium-ion batteries, based on perfluoropolyethers (PFPEs). In a paper published in the Proceedings of the National Academy of Sciences, the team reports that these electrolytes exhibit thermal stability beyond 200° C and a transference number of at least 0.91 (more than double that of conventional electrolytes).
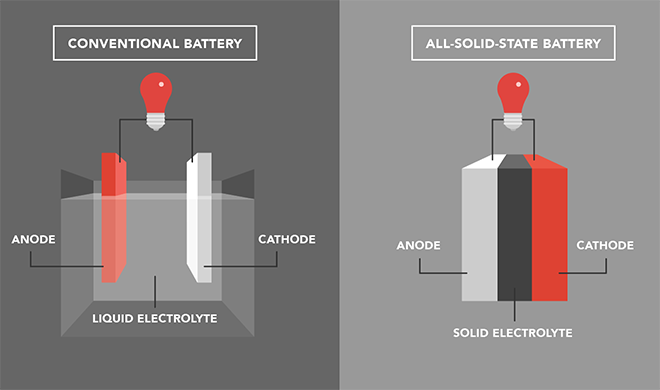
Most commercial lithium-ion batteries contain an electrolyte that is dissolved in flammable organic solvents. Overheating, overcharging or damage to the battery can result in fire. Various researchers have found alternative non-flammable electrolytes, but these alternatives compromised performance.
“In addition to being non-flammable, PFPE exhibits very interesting properties such as its ion transport,” said the study’s lead author Dominica Wong. “That makes this electrolyte stand apart from previous discoveries.”
“There is a big demand for these batteries and a huge demand to make them safer,” said Professor Joseph DeSimone of the University of North Carolina at Chapel Hill. “Researchers have been looking to replace this electrolyte for years, but nobody had ever thought to use this material called perfluoropolyether, or PFPE, as the main electrolyte material in lithium-ion batteries before.”
The discovery began when DeSimone realized that PFPE, a material that he had been researching for the Office of Naval Research to prevent marine life from sticking to the bottom of ships, had a similar chemical structure to a polymeric electrolyte commonly studied for lithium-ion batteries. Nitash Balsara, of Lawrence Berkeley National Laboratory, and his team were then tasked with studying lithium-ion transport within the electrolyte and found compatible electrodes to assemble a battery.
“When we discovered that we could dissolve lithium salt in this polymer, that’s when we decided to roll with it,” said Wong. “Most polymers don’t mix with salts, but this one did—and it was non-flammable. It was an unexpected result.”
The team will now focus on optimizing electrolyte conductivity and improving battery cycling characteristics, which are necessary before the new material can be scaled up for use in commercial batteries.
Source: Proceedings of the National Academy of Sciences via Green Car Congress