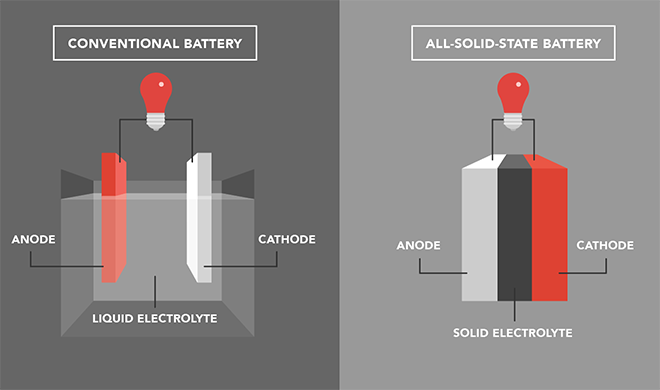
Researchers at the DOE’s Oak Ridge National Laboratory have developed a unique battery chemistry that promises to greatly extend battery life by making the electrolyte serve two functions.
In a study, Pushing the Theoretical Limit of Li-CFx Batteries: A Tale of Bi-functional Electrolyte, which was published in the Journal of the American Chemical Society, ORNL researchers described an electrolyte that serves not only as an ion conductor but also as a cathode supplement. This cooperative chemistry, enabled by the use of an ORNL-developed solid electrolyte, delivers an extra boost to the battery’s capacity and extends the lifespan of the device.
“This bi-functional electrolyte revolutionizes the concept of conventional batteries and opens a new avenue for the design of batteries with unprecedented energy density,” said ORNL’s Chengdu Liang.

The team demonstrated the concept in a lithium carbon fluoride battery. When the researchers incorporated a solid lithium thiophosphate electrolyte, the battery generated a 26 percent higher capacity than what its theoretical maximum would be if each component acted independently. The increase, explains Liang, is caused by the cooperative interactions between the electrolyte and cathode.
“As the battery discharges, it generates a lithium fluoride salt that further catalyzes the electrochemical activity of the electrolyte,” Liang said. “This relationship converts the electrolyte – conventionally an inactive component in capacity – to an active one.”
The improvement in capacity could translate into years or even decades of extra life, depending on how the battery is engineered and used.
Source: Science Daily
Image courtesy of DOE/Oak Ridge National Laboratory