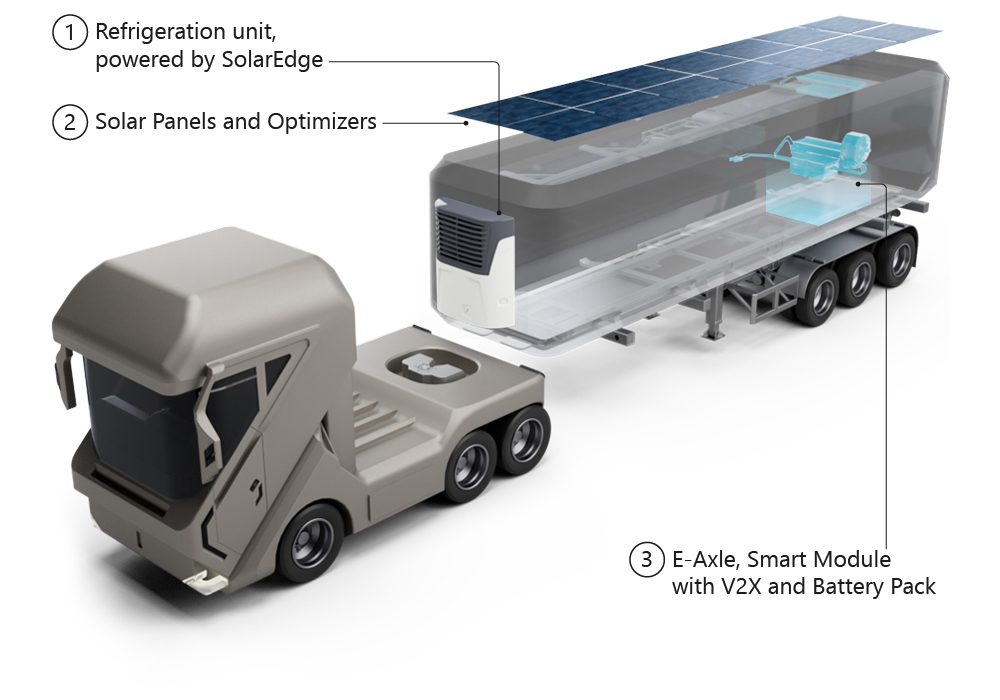
The process uses silicon to replace graphite as an element in Li-ion cells, which could lead to longer-lasting, more powerful batteries.
The race is on to build a better battery – it seems we’re reading about new advances every week. The latest breakthrough comes from researchers at Rice University and Lockheed Martin, who’ve found a way to make multiple high-performance anodes from a single silicon wafer.
The process uses silicon to replace graphite as an element in Li-ion cells, which could lead to longer-lasting, more powerful batteries. In a previous advance, the research team found that porous silicon soaked up 10 times more lithium than graphite.
The new work, which was published this month in the journal Chemistry of Materials, details the process by which Swiss cheese-like silicon “sponges” that store more than four times their weight in lithium can be electrochemically lifted off of wafers.
Because silicon expands as it absorbs lithium ions, the sponge-like configuration gives it room to grow internally without degrading the battery’s performance, the researchers reported. They discovered that the electrochemical etching process used to create the pores can also separate the sponge from the substrate, which is then reused to make more sponges.
The product is a tough film that can be attached to a current collector (in this case, a thin layer of titanium on copper) and placed in a battery configuration. The team built a working lithium-ion battery with a discharge capacity of 1,260 milliamp-hours per gram, a capability that remained consistent through 20 cycles. The next step is to vastly increase the number of charge-discharge cycles, a critical feature for commercial applications in which rechargeable batteries are expected to last for years.
Source: Rice University via Green Car Congress
Image Credit: ACS, Thankur et al.