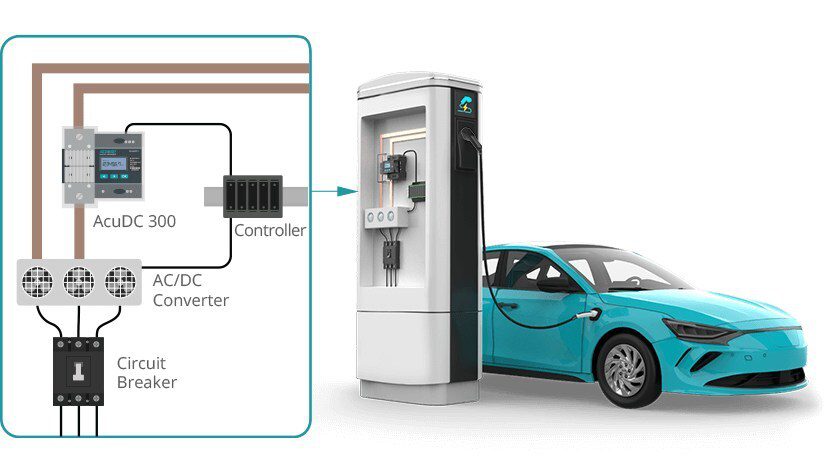
New research from MIT could lead to batteries that pack more power per pound and last longer. A new paper co-authored by Yuming Chen and Ziqiang Wang at MIT explores the idea of using pure lithium as the battery’s anode.
The design is part of a concept for developing safe solid-state batteries by dispensing with the liquid or polymer gel usually used as the electrolyte material between a battery’s two electrodes. Solid-state electrolytes are expected to be safer than volatile liquid electrolytes, which can explode under extreme conditions.
Although many other groups are working on what they call solid batteries, Li says most of those systems actually work better with some liquid electrolyte mixed with the solid electrolyte material. “In our case it’s truly all solid,” he says. “There is no liquid or gel in it of any kind.”
“There has been a lot of work on solid-state batteries, with lithium metal electrodes and solid electrolytes,” Li says, but these efforts have faced a number of issues. One problem is that none of the proposed solid electrolytes are chemically stable while in contact with the highly reactive lithium metal, so they tend to degrade over time. Most attempts to overcome this have focused on designing solid electrolyte materials that are stable against lithium metal, which turns out to be difficult. Instead, Li and his team adopted an unusual design that utilizes two additional classes of solids, “mixed ionic-electronic conductors” (MIEC) and “electron and Li-ion insulators” (ELI), which the engineers say are chemically stable in contact with lithium metal.
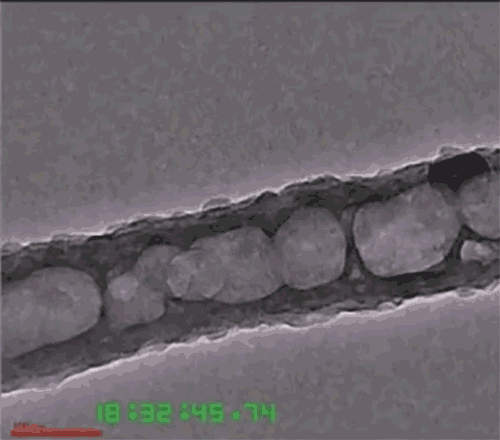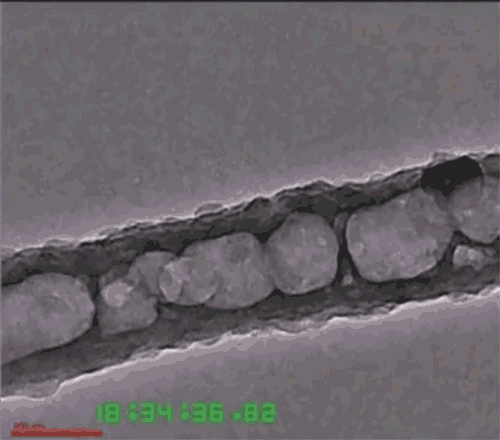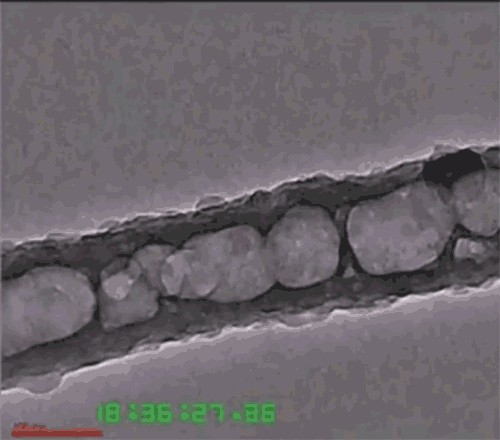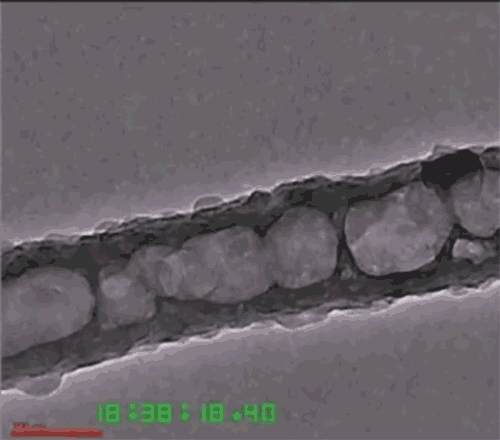
Another issue is that the lithium anode expands and contracts with each charge-discharge cycle, eventually causing the solid electrolytes to fracture or detach. To deal with that, the researchers developed a three-dimensional nanoarchitecture in the form of a honeycomb-like array of hexagonal MIEC tubes, partially infused with the solid lithium metal to form one electrode of the battery, but with extra space left inside each tube. The honeycomb structure allows the lithium to expand and contract without changing the electrode’s outer dimensions or the boundary between the electrode and electrolyte. The other material, the ELI, serves as a mechanical binder between the MIEC walls and the solid electrolyte layer.
Because the honeycomb-like walls are made of chemically stable MIEC, the lithium never loses electrical contact with the material, Li says. Thus, the battery can remain mechanically and chemically stable as it goes through its cycles. The team has proved the concept experimentally, putting a test device through 100 cycles of charging and discharging without fracturing any of the solids.
The new system could lead to anodes that weigh one fourth as much as their conventional counterparts for the same amount of storage capacity. Combined with new designs for lightweight cathodes, this could result in substantially lighter batteries. For example, the team hopes it could lead to cellphones that could be charged just once every three days, without making the phones any heavier or bulkier.
The team has only built small lab-scale devices so far, but Li thinks that the technology can be scaled up very quickly. The materials needed, mostly manganese, are significantly cheaper than the nickel or cobalt used by other systems, so these cathodes could cost as little as a fifth as much as current versions.
Source: MIT