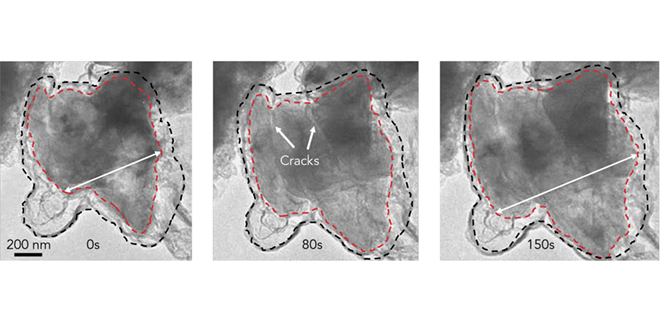
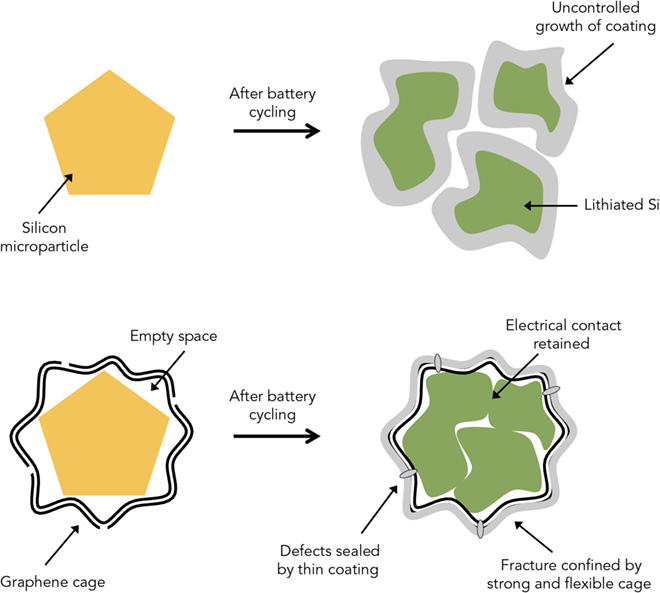
Battery boffins are buzzing about silicon anodes, which could store up to 10 times more energy than today’s anodes. The catch is that silicon particles tend to swell and crack during cycling, and to react with the electrolyte in unwanted ways.
Now a team from Stanford University and the Department of Energy’s SLAC National Accelerator Laboratory has found a way to solve these problems – by encasing each silicon particle in a custom-fit graphene cage.
In “Growth of conformal graphene cages on micrometre-sized silicon particles as stable battery anodes,” published in Nature Energy, the researchers describe a method for building microscopic graphene cages big enough to let the silicon particle expand, but small enough to hold the pieces together when the particle falls apart. The cages also block undesired chemical reactions with the electrolyte.
“In testing, the graphene cages actually enhanced the electrical conductivity of the particles and provided high charge capacity, chemical stability and efficiency,” said research leader Yi Cui. “The method can be applied to other electrode materials, too.”
Cui and his colleagues had already found ways to confine and protect silicon nanoparticles, but the efficiency of the resulting anodes was not high enough, and nanoparticles are expensive and hard to manufacture.
“This new method allows us to use much larger silicon particles that are one to three microns, or millionths of a meter, in diameter, which are cheap and widely available,” Cui said. “In fact, the particles we used are very similar to the waste created by milling silicon ingots to make semiconductor chips; they’re like bits of sawdust of all shapes and sizes.”
To build graphene cages around silicon particles, researchers coated the particles with nickel, grew layers of graphene on top of the nickel, then used acid to dissolve the nickel away, leaving enough space for the silicon to expand inside the cage.
Now the team will work on fine-tuning the process, Li added, and on producing caged silicon particles in large enough quantities to build commercial-scale batteries for testing.
Source: SLAC National Accelerator Laboratory
Image: Y. Li et al., Nature Energy