Carbon nanotubes could make high-powered, fast-charging lithium metal batteries an alternative to lithium-ion batteries, according to new research from Rice University.
In a paper published this month in the journal Advanced Materials, the researchers show how a carbon nanotube film can be used to keep batteries safe from dendrites, spear-like protrusions that naturally form on unprotected lithium metal anodes. Left unchecked, dendrites can penetrate the battery’s core, pierce the cathode, and cause it to fail.
Lithium metal’s advantages over lithium-ion electrodes include faster charging and the potential for as much as ten times greater energy density. However, the risk of battery failure caused by those pesky dendrites has limited lithium metal’s commercial use. By coating the lithium metal with a carbon nanotube film, the Rice researchers say they’ve mitigated the growth of dendrites over 580 charging cycles, while maintaining the material’s Faraday efficiency at 99.8 percent.
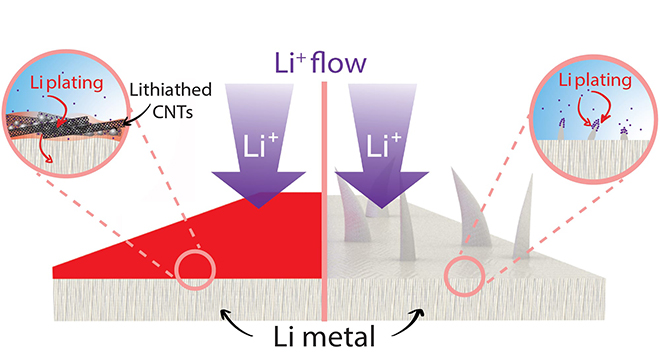
Illustration shows how lithium metal anodes developed at Rice University are protected from dendrite growth by a film of carbon nanotubes. Courtesy of the Tour Group
“One of the ways to slow dendrites in lithium-ion batteries is to limit how fast they charge,” said James Tour, a synthetic organic chemist whose Rice lab conducted the study. “People don’t like that. They want to be able to charge their batteries quickly. What we’ve done turns out to be really easy. You just coat a lithium metal foil with a multi-walled carbon nanotube film. The lithium dopes the nanotube film, which turns from black to red, and the film in turn diffuses the lithium ions.”
While the battery is in use, the carbon nanotube film discharges its stored ions, which are then refilled by the lithium anode underneath, enabling the film to continue mitigating dendrite growth.
“Physical contact with lithium metal reduces the nanotube film, but balances it by adding lithium ions,” said Rodrigo Salvatierra, a Rice postdoctoral researcher and co-lead author of the paper. “The ions distribute themselves throughout the nanotube film.”
Source: Rice University