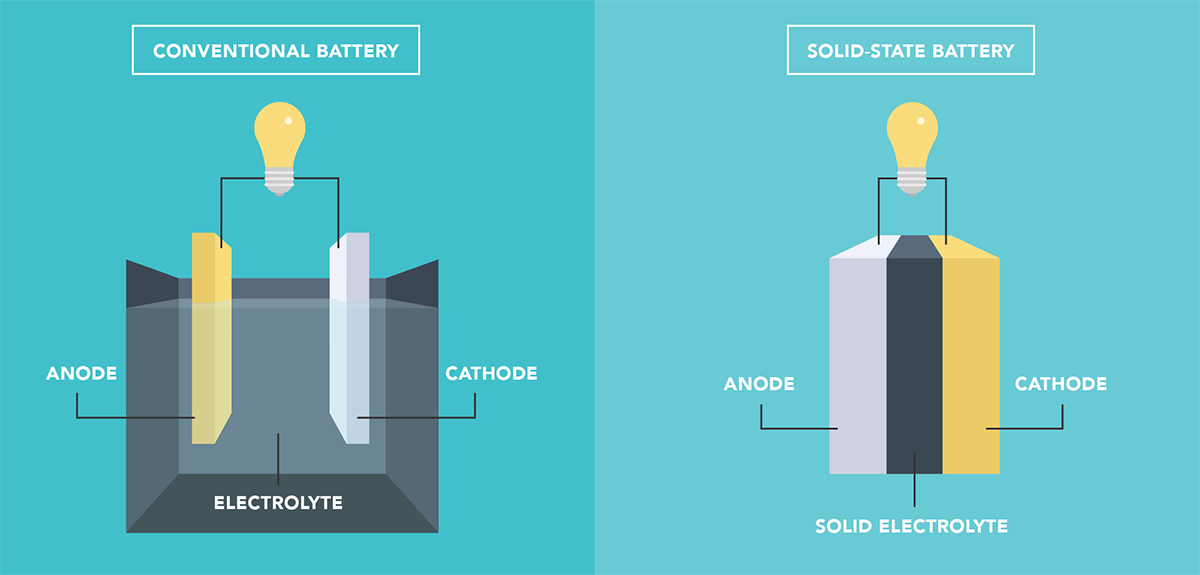
Many in the EV industry see solid-state batteries as the future, and one of the reasons is that the more stable solid electrolyte is expected to be safer than current liquid electrolytes. However, new research indicates that the picture may be more complex.
A recent study led by the DOE’s Sandia National Laboratories, published in the scientific journal Joule, found that a solid electrolyte can still fail under certain circumstances, such as when the battery is crushed, punctured, or when built-up pressure causes a reaction between internal oxygen and lithium.
When the solid electrolyte does fail, the resulting short circuit could cause temperatures to rise as much as they would in a conventional lithium-ion battery failure, potentially igniting surrounding materials.
Whether it has a solid electrolyte or not, any battery that contains lithium is also vulnerable to dendrite spikes—a problem of this kind recently forced GM to recall almost all of its Chevrolet Bolts to replace the battery packs.
Solid-state batteries still have the potential to be safer and more energy-dense than conventional lithium-ion cells, Alex Bates, a Sandia postdoctoral researcher who led the study, said in an interview with ScienceDaily. He says adding a small amount of liquid electrolyte could “greatly increase performance while only having a small impact on safety.”
The researchers considered solid-state batteries that include a relatively small amount of liquid electrolyte, which is often added at the cathode to reduce interfacial resistance. “While the addition of small amounts of liquid electrolyte increases heat release under specific failure scenarios, it may be small enough that other considerations, such as manufacturability and performance, are more important commercially,” reads a summary of the findings. “We show that short-circuited all-solid-state batteries can reach temperatures significantly higher than conventional Li-ion, which could lead to fire through flammable packaging and/or nearby materials.”
“Polymer solid electrolytes with sufficient shear modulus are theorized to block dendritic growth and prevent short circuits,” the researchers write. “However, this theory is not applicable to ceramic solid electrolytes, where grain boundaries offer growth pathways, and fracture is a critical failure mode. Indeed, many studies show that Li dendrites can grow through solid electrolytes and short-circuit the cells.”
“In addition to challenges with dendrite-induced short circuits, solid-state cells also suffer from high interfacial resistance between solid electrolytes and active cathode materials. This problem can be addressed by adding some liquid electrolyte to the solid electrolyte cathode region, greatly reducing interfacial resistance.”
The researchers note that other solutions, such as surface coatings or surface structure designs, tend to increase manufacturing costs, so a solid-state cell with a small amount of liquid electrolyte may be the best way to go. “Our results suggest that adding a small enough amount of liquid electrolyte may be a reasonable step toward the commercialization of SSBs, even when safety is considered.”
Source: Joule via Green Car Reports