A set of three recent studies funded by the Korean government investigated a family of lithium-ion batteries composed of nickel, cobalt and aluminum. NCA batteries offer high energy density, but degrade significantly with each charge/discharge cycle. The high heat of vehicle environments can intensify the degradation, and even cause complete battery failure.
Researchers at the Brookhaven National Lab’s Center for Functional Nanomaterials (CFN) used x-ray imaging and electron microscopy to get a detailed picture of the NCA battery’s electrochemical reactions.

“The relationship between structural changes and the catastrophic thermal runaway impacts both safety and performance,” said Brookhaven physicist Xiao-Qing Yang. “The in-depth understanding of that relationship will help us develop new materials and advance this NCA material to prevent that dangerous degradation.”
“After each cycle of charge/discharge, we saw the atomic structure transition from uniform crystalline layers into a disordered rock-salt configuration,” said Brookhaven scientist Eric Stach. “During this transformation, oxygen leaves the destabilized battery compound. This excess oxygen, leached at faster and faster rates over time, actually contributes to the risk of failure and acts as fuel for a potential fire.”
The first study, published in Chemistry of Materials, explored the NCA battery using x-ray diffraction and spectroscopy techniques. “We were able to test the battery cycling in situ, meaning we could watch the effects of increasing heat in real time,” said Brookhaven chemist and study coauthor Seong Min Bak. “We pushed the fully charged NCA coin-cell battery out of thermal equilibrium by heating it all the way to 500 degrees Celsius.”
As the temperature rose, x-rays struck the sample and revealed the widespread transition from one crystal structure to another. The team also measured the amount of oxygen and carbon dioxide released by the NCA sample.
“The oxygen release peaked between 300 and 400 degrees Celsius during our trials, which is above the operating temperature for most vehicles,” Bak said. “But that temperature threshold dropped for a highly charged battery, suggesting that operating at full energy capacity accelerates structural degradation and vulnerability.”
The next study, also published in Chemistry of Materials, used transmission electron microscopy (TEM) to pinpoint the effect of an initial charge on the battery’s surface structure. The highly focused electron beams available at CFN revealed individual atom positions as an applied current pushed pristine batteries to an overcharged state.
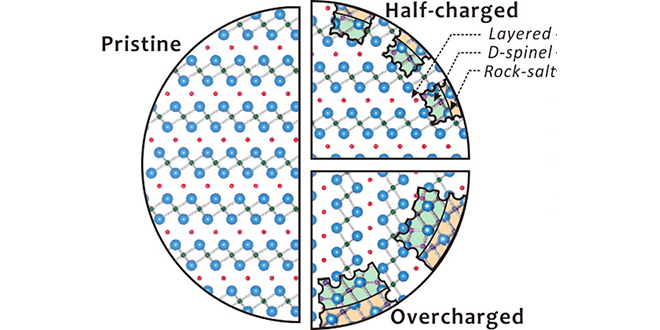
“The surface changes matched the rock-salt evolution found in the x-ray study,” said study coauthor Sooyeon Hwang. “Even with just one charge on the NCA battery we saw changes in the crystalline structure, and it grew much worse as the charge level increased.”
The final study, published in Applied Materials and Interfaces, used in situ electron microscopy to track the heat-driven decomposition of NCA materials at different states of charge. Even though pristine and uncharged NCA samples remained stable up to 400 degrees Celsius, charging introduced the usual decomposition and vulnerabilities. The full story, however, was more nuanced.

“We saw the same overall degradation patterns, but the real-time TEM revealed an unexpected twist within individual particles,” Stach said. “When fully charged, some particles released oxygen and began to shift toward disorder down at temperatures below 100 degrees Celsius – definitely plausible for a lithium-ion battery’s normal operation.”
Added Hwang, “Those unstable, degraded particles may trigger the chain reaction of so-called thermal runaway at lower temperatures than expected, and that free oxygen would feed the fire springing from an overheated battery.”
“We plan to push these investigative techniques even further to track the battery’s structure in real-time as it charges and discharges under real operating conditions – we call this in operando,” Stach said. “Brookhaven’s National Synchrotron Light Source II will be a game-changer for this kind of experimentation, and I’m eager to take advantage of that facility’s ultra-bright x-rays to track internal and surface evolutions in these materials.”
Source: Brookhaven National Laboratory




















































































