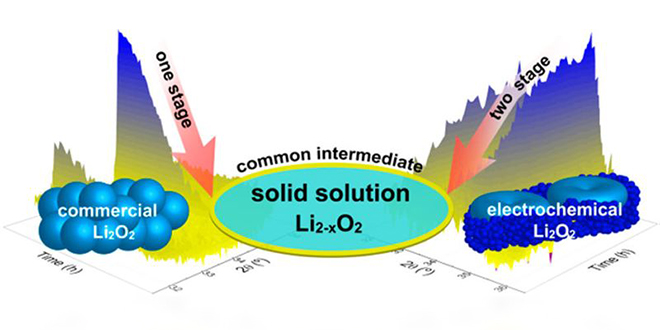
A team from Delft University in the Netherlands and the University of Waterloo in Canada has used operando X-ray diffraction to show that oxidation of electrochemically generated lithium peroxide (Li2O2) in high-energy-density Li-air batteries occurs in two stages, but in only one step for commercial (crystalline) Li2O2.
In a paper published in the Journal of the American Chemical Society, the authors conclude that their findings not only reveal the fundamental nature of the charge reaction in Li-air batteries, but also show the impact that the nature of the lithium peroxide has on the oxidation mechanism. Controlling this process may be the key to high-performance Li-air batteries.
“Research into nonaqueous Li-air batteries has exploded over the past few years,” writes Swapna Ganapathy and his co-authors. “This is primarily due to their high theoretical specific energy in the charged state (∼11,500 Wh/kg), making them extremely attractive for electric automobiles. Several bottlenecks that impede the functioning of this battery system need to be addressed before it can become viable.”
“Of fundamental importance is understanding the mechanism of lithium peroxide formation and oxidation. Over the past years there has been significant progress in the understanding of the process…but the mechanism of Li2O2 oxidation remains less well understood, with the elusive LiO2 superoxide intermediate remaining experimentally very difficult to observe.
“In this article, we combine operando X-ray diffraction, Rietveld refinement, calculations, and online electrochemical mass spectrometry to elucidate the mechanism of the oxidation evolution reaction, comparing electrochemically generated Li2O2 and bulk crystalline (commercial) Li2O2 during the charge reaction. A clear difference is observed, which can be explained by the difference in the nature of the particles and crystallites.”
Source: Journal of the American Chemical Society via Green Car Congress
Image: ACS, Ganapathy et al.